 Repulsion by bonding pairs at 120° is much smaller and less important. It includes the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that seesaw. An equatorial lone pair is repelled by only two bonding pairs at 90°, whereas a hypothetical axial lone pair would be repelled by three bonding pairs at 90° which would make it stable. Part A Below is a cartoon drawing of a seesaw molecular geometry which is based upon the trigonal bipyramidal electron group geometry. This is true because the lone pair occupies more space near the central atom (A) than does a bonding pair of electrons. The T shape and trigonal bipyramidal geometry of iodine trichloride lead to a bond angle (Cl-I-Cl) slightly smaller than 90 ° and greater than. lAnswer : (a) Molecular structure Square planar Bond angle (b). The molecular geometry and shape of the molecule can be predicted from the following table: General formula: Number of bond pairs (B) Number of lone pairs (E) Molecular geometry: Shape: AX: 1: 0. An atom bonded to 5 other atoms (and no lone pairs) forms a trigonal bipyramid with two axial and three equatorial positions, but in the seesaw geometry one of the atoms is replaced by a lone pair of electrons, which is always in an equatorial position. With one lone pair on this electronic geometry, the lone pair must occupy an equatorial position and the molecular geometry becomes see-saw shape. Seesaw Determine the molecular geometry about each interior atom and make a sketch. Question: A molecule with a seesaw molecular geometry has a bond angle of A) <120 degree for equatorial bonds and < 90 degree for axial bonds. The angle formed by the F-S-F axial plane is 173 degrees and 3.28 Angstroms in length, which can be attributed to the lone pair of electrons on the S atom. 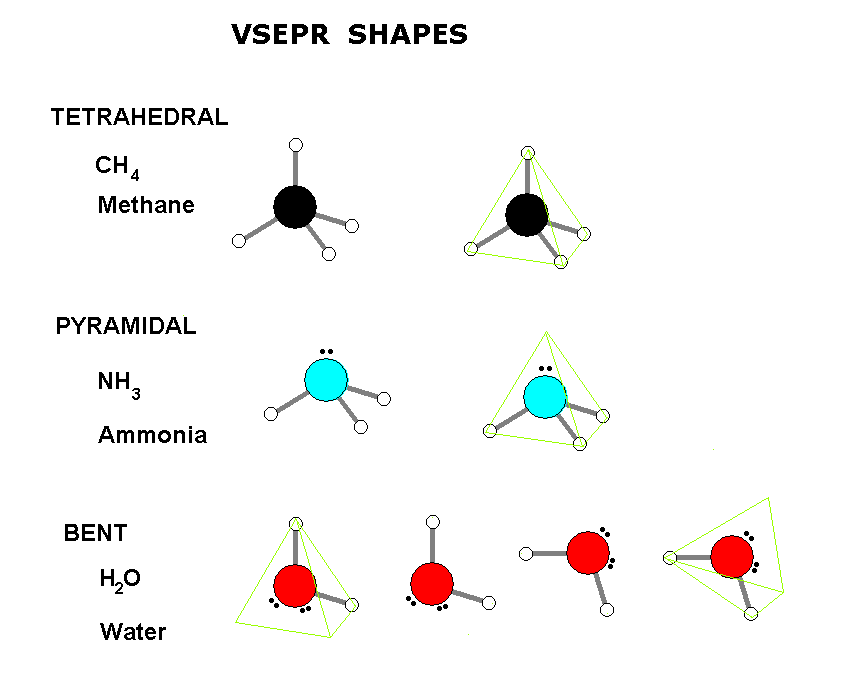
Sulfur is the central atom, two fluorine atoms are on the equatorial plane, and two are on the axial plane. 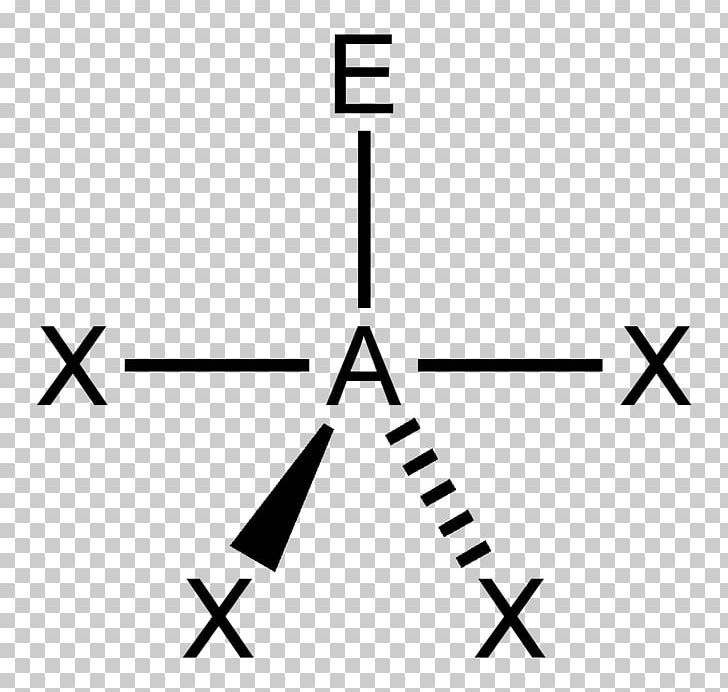 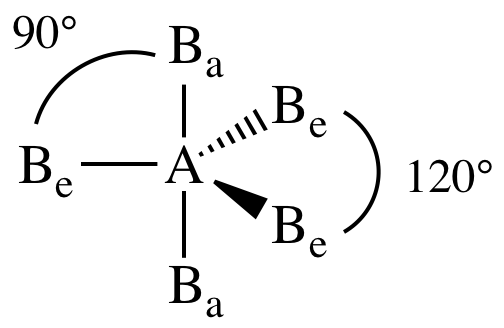
The seesaw geometry occurs when a molecule has a steric number of 5, with the central atom being bonded to 4 other atoms and 1 lone pair (AX 4E 1 in AXE notation). An example of a seesaw shaped molecule is sulfur tetrafluoride, or SF4. Quick facts: Seesaw molecular geometry, Examples, Point gr. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
